
Publications
2025
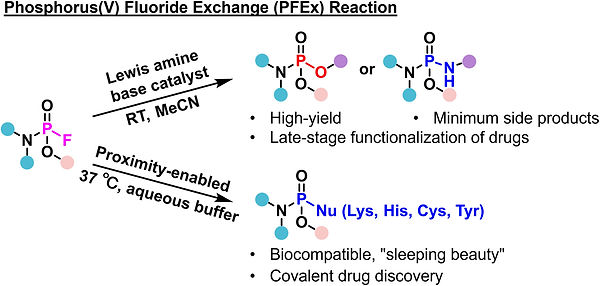
Yang, S.; Li, Y.; Yang, L.; Yu, B.* Phosphorus Fluoride Exchange Chemistry: An Emerging Strategy for Covalent Drug Discovery. Tetrahedron Chem. https://doi.org/10.1016/j.tchem.2025.100143
Wang, S.; Faucher, F.; Bertolini, M., Kim, H.; Yu, B.; Cao, L.; Roeltgen, K.; Lovell, S.; Shanker, V.; Boyd, S.; Wang, L.; Bartenschlager, R.; Bogyo, M. Identification of covalent cyclic peptide inhibitors targeting protein–protein interactions using phage display. Journal of the American Chemical Society. 2025, 147, 7461−7475.

2024
Cao, L.; Yu, B.; Klauser, P.; Zhang, P.; Li, S.; Wang, L.* Arginine Accelerates Sulfur Fluoride Exchange and Phosphorus Fluoride Exchange Reactions between Proteins. Angewandte Chemie International Edition, 2024. DOI: https://doi.org/10.1002/anie.202412843

Yang, L.; Yuan, Z.; Li, Y.; Yang, S.; Yu, B.* Sulfur (VI) fluoride exchange (SuFEx): a versatile tool to profile protein-biomolecule interactions for therapeutic development. Medicinal Chemistry Research, 2024. DOI: https://doi.org/10.1007/s00044-024-03255-7
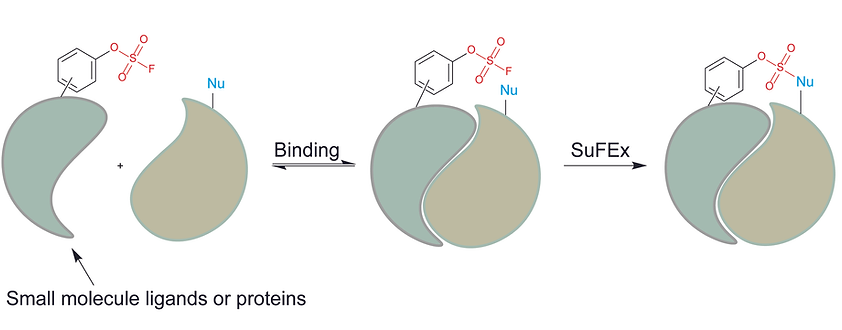
Cao, L.; Yu, B.; Li, S.; Zhang, P.; Li, Q.; Wang, L. Genetically enabling phosphorus fluoride exchange click chemistry in proteins. Chem, 2024. DOI: https://doi.org/10.1016/j.chempr.2024.02.010

2023
Yu, B.; Yang, X.; Yuan, Z.; Wang, B., Prodrugs of sulfide and persulfide species: Implications in their different pharmacological activities. Current Opinion in Chemical Biology, 2023, 75:102329.

Yu, B.; Cao, L.; Li, S.; Klauser, P.; Wang, L. Proximity-Enabled Sulfur Fluoride Exchange Reaction in Protein Context. Chemical Science , 2023, 14, 7913-7921. DOI: https://doi.org/10.1039/D3SC01921G

Klauser, P.; Chopra, S.; Cao, L.; Bobba, K.; Yu, B.; Seo, Y.; Chan, E.; Flavell, R.; Michael, E.; Wang, L. Covalent Proteins as Targeted Radionuclide Therapies Enhance Antitumor Effects. ACS Central Science, 2023, 9, 6, 241–1251 . DOI: 10.1021/acscentsci.3c00288

Li, S.; Wang, N.; Yu, B.; Sun, W.; Wang, L. Genetically Encoded Chemical Cross-linking of Carbohydrate. Nature Chemistry, 2023,15,33–42. DOI: https://doi.org/10.1038/s41557-022-01059-z

Sun, W.; Wang, N.; Liu, H.; Yu, B.; Jin, L.; Ren, X.; Shen, Y.; Wang, L. Genetically Encoded Chemical Cross-linking of RNA in vivo. Nature Chemistry, 2023, 15,21–32. DOI: https://doi.org/10.1038/s41557-022-01038-4

2022
Yu, B.; Li, S.; Tabata, T.; Wang, N.; Li, C.; Kumar, R.; Sun, W.; Liu, J.; Ott, M.; Wang, L. Accelerating PERx Reaction Enables Covalent Nanobodies for Potent Neutralization of SARS-CoV-2 and Variants.
Chem. 2022, 8, 2766-2783. DOI: https://doi.org/10.1016/j.chempr.2022.07.012

Yu, B.; Kang, T.; Xu, Y.; Liu, Y.; Ma, Y.; Ke, B. Prodrugs of Persulfide and Sulfide: Is There a Pharmacological Difference between the Two in the Context of Rapid Exchanges among Various Sulfur Species in vivo? Angewandte Chemie International Edition. 2022, 61, e202201668. (Front cover). DOI: https://doi.org/10.1002/anie.202201668

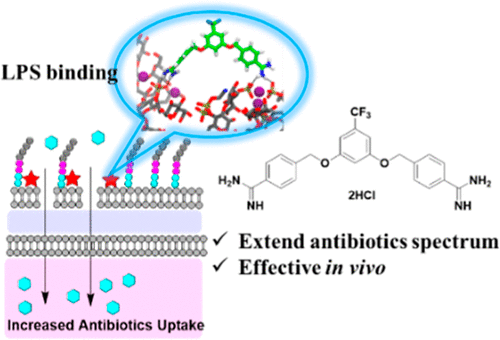
Yu, B.; Choudhury, M.; Yang, X.; Benoit, S.; Womack, E.; Lyles, K; et al. Restoring and Enhancing the Potency of Existing Antibiotics against Drug-Resistant Gram-Negative Bacteria through the Development of Potent Small-Molecule Adjuvants. ACS Infectious Diseases. 2022, 8, 1491–1508. DOI:https://doi.org/10.1021/acsinfecdis.2c00121
Yu, B.; Yuan, Z.; Wang, B. Persulfide Prodrugs. in Pluth, M. & Wang, B. (Eds.), Hydrogen Sulfide: Chemical Biology Basics, Detection Methods, Therapeutic Applications, and Case Studies. John Wiley & Sons, Hoboken, NJ, 2022. (Book chapter)
2021
Liu, J.; Cao, L.; Klauser, P.; Cheng, R.; Berdan, V.; Sun, W.; Wang, N.; Ghelichkhani, F.; Yu, B.; Rozovsky, S.; Wang, L. A Genetically Encoded Fluorosulfonyloxybenzoyl-l-lysine for Expansive Covalent Bonding of Proteins via SuFEx Chemistry. Journal of the American Chemical Society. 2021, 143, 10341–10351. DOI: https://doi.org/10.1021/jacs.1c04259

Bakalarz, D.; Korbut, E.; Yuan, Z.; Yu, B.; Wójcik, D.; Danielak, A.; et al. Novel Hydrogen Sulfide (H2S)-Releasing BW-HS-101 and Its Non-H2S Releasing Derivative in Modulation of Microscopic and Molecular Parameters of Gastric Mucosal Barrier. International Journal of Molecular Sciences. 2021, 22, 5211. DOI: https://doi.org/10.3390/ijms22105211
2020
Yu, B.; Yuan, Z.; Yang, X.; Wang, B. Prodrugs of Persulfides, Sulfur Dioxide, and Carbon Disulfide: Important Tools for Studying Sulfur Signaling at Various Oxidation States. Antioxidants & Redox signaling. 2020, 33, 1046-1059. DOI: https://doi.org/10.1089/ars.2019.7880
Li, S.; Yang, B.; Kobayashi, T.; Yu, B.; Wang, L. Genetically Encoding Thyronine for Fluorescent Detection of Peroxynitrite. Bioorganic & Medicinal Chemistry. 2020, 18, 115665. DOI: https://doi.org/10.1016/j.bmc.2020.115665

Yang, X.; Pan, Z.; Choudhury, M.R.; Yuan, Z.; Anifowose, A.; Yu, B.; Wang, W.; Wang, B. Making Smart Drugs Smarter: The Importance of Linker Chemistry in Targeted Drug Delivery. Medicinal Research Reviews. 2020, 40, 2682-2713. DOI: https://doi.org/10.1002/med.21720
2019
Liu, L.; Yu, B.; Han, M; Yuan, S; Wang, N. Mild Cognitive Impairment Understanding: An Empirical Study by Data-Driven Approach. BMC Bioinformatics. 2019, 20, 481.DOI: https://doi.org/10.1186/s12859-019-3057-1
Ji, X.; Pan, Z.; Zheng, Y.; Yu, B.; De La Cruz, L. K.; Zheng, Y.; Ke, B.; Wang, B. Click and Release: Bioorthogonal Approaches to “On-demand” Activation of Prodrugs. Chemical Society Reviews. 2019, 48, 1077-1094. DOI: https://doi.org/10.1039/C8CS00395E
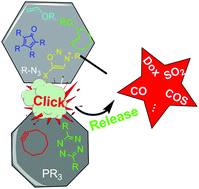
Ji, X.; Aghoghovbia, R. E.; De La Cruz, L. K.; Pan, Z.; Yang, X.; Yu, B.; Wang, B. Click and Release: A High-Content Bioorthogonal Prodrug with Multiple Outputs. Organic Letters. 2019, 21, 3649-3652. DOI:https://doi.org/10.1021/acs.orglett.9b01086
2018
Yu, B.; Zheng, Y.; Yuan, Z.; Li, S.; Zhu, H.; De La Cruz, L. K.; Zhang, J.; Ji, K.; Wang, S.; Wang, B. Toward Direct Protein S-Persulfidation: A Prodrug Approach That Directly Delivers Hydrogen Persulfide. Journal of the American Chemical Society. 2018, 140, 30–33. DOI: https://doi.org/10.1021/jacs.7b09795

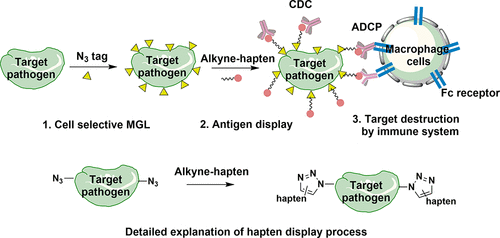
Li, S.; Yu, B.; Wang, J.; Zheng, Y.; Zhang, H.; Walker, M. J.; et al. Biomarker-based Metabolic Glycan Labelling for Cancer Cell Imaging and Enhanced Immune Response. ACS Chemical Biology. 2018, 13, 1686–1694. DOI: https://doi.org/10.1021/acschembio.8b00350
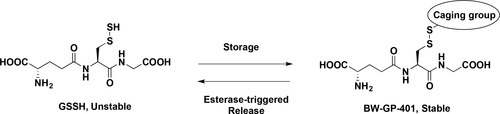
Yuan, Z.; Zheng, Y.; Yu, B.; S, Wang.; Yang, X.; Wang, B. Esterase-Sensitive Glutathione Persulfide. Organic Letters. 2018, 20, 6364-6367. DOI: https://doi.org/10.1021/acs.orglett.8b02611
Zheng, Y.; Ji, X.; Yu, B.; Ji, K.; Gallo, D.; Csizmadia, E.; et al. Enrichment-Triggered Prodrug Activation Demonstrated Through Mitochondria-Targeted Delivery of Doxorubicin and Carbon Monoxide. Nature Chemistry. 2018, 10, 787–794. DOI: https://doi.org/10.1038/s41557-018-0055-2


Zheng, Y.; Yu, B.; De La Cruz, L. K.; Choudhury, M.R.; Chittavong, V.; Anifowose, A.; Wang, B. Toward Hydrogen Sulfide Based Therapeutics: Critical Drug Delivery and Developability Issues. Medicinal Research Reviews. 2018, 38, 57-100. DOI: https://doi.org/10.1002/med.21433
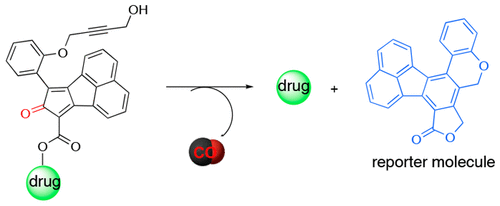
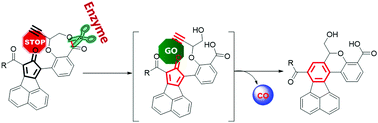
De La Cruz, L. K.; Benoit, S. L.; Pan, Z.; Yu, B.; Maier, R. J.; Ji, X.; Wang, B. Click, Release, and Fluoresce: A Chemical Strategy for a Cascade Prodrug System for Codelivery of Carbon Monoxide, a Drug Payload, and a Fluorescent Reporter. Organic Letters. 2018, 20, 897-900. DOI: https://doi.org/10.1021/acs.orglett.7b03348
2015 to 2017
Zheng, Y.; Yu, B.; Li, Z.; Yuan, Z.; Organ, C. L.; Trivedi, R. K.; Wang, S.; Lefer, D. J.; Wang, B. An Esterase-Sensitive Prodrug Approach for Controllable Delivery of Persulfide Species. Angewandte Chemie International Edition. 2017, 56, 11749 –11753. DOI: https://doi.org/10.1002/ange.201704117

Ji, X.; Ji, K.; Chittavong, V.; Yu, B.; Pan, Z.; Wang, B. An Esterase-Activated Click and Release Approach to Metal-Free CO-Prodrugs. Chemical Communications. 2017, 53, 8296-8299. DOI: https://doi.org/10.1039/C7CC03832A
Li, Z.; Organ, C. L.; Trivedi, R. K.; Zheng, Y.; Yu, B.; Wang, B. Lefer, D. J. A Novel Hydrogen Sulfide Prodrug, BW-HP-202, Attenuates Myocardial Ischemia/Reperfusion Injury. The FASEB Journal. 2017, 31, 544-544. DOI: https://doi.org/10.1096/fasebj.31.1_supplement.lb544
Zheng, Y.; Yu, B.; Ji, K.; Pan, Z.; Chittavong, V.; Wang, B. Esterase-Sensitive Prodrugs with Tunable Release Rates and Direct Generation of Hydrogen Sulfide. Angewandte Chemie International Edition. 2016, 55, 4514-4518. DOI: https://doi.org/10.1002/anie.201511244

Ji, X.; Damera, K.; Zheng, Y.; Yu, B.; Otterbein, L. E.; Wang, B. Toward Carbon Monoxide-Based Therapeutics: Critical Drug Delivery and Developability Issues. Journal of Pharmaceutical Sciences. 2016, 105, 406-416. DOI :https://doi.org/10.1016/j.xphs.2015.10.018
Li, S.; Zhu, H.; Wang, J.; Wang, X.; Li, X.; Ma, C.; Wen, L.; Yu, B.; Wang, Y.; Li, J.; Wang, P. G. Comparative Analysis of Cu (I)-Catalyzed Alkyne-Azide Cycloaddition (CuAAC) and Strain-Promoted Alkyne-Azide Cycloaddition (SPAAC) in O-GlcNAc Proteomics. Electrophoresis. 2016, 37, 1431–1436. DOI: https://doi.org/10.1002/elps.201500491
Damera, K.; Yu, B.; Wang, B. Stereoselective Synthesis of 1-Methyl-3´,4´,5´,6´- tetrahydrospiro[indoline-3,2´-pyran]-2-one Derivatives via Prins Cyclization. The Journal of Organic Chemistry. 2015, 80, 5457-5463. DOI: https://doi.org/10.1021/acs.joc.5b00249